Philips CPAP Machine Recall
The U.S. Food and Drug Administration (FDA) issued a recall notice concerning several Philips Respironics CPAP machines in June 2021. Philips manufactured the problematic devices between 2009 and April 26, 2021, before issuing a voluntary recall for its widely-used medical devices that assist patients with complications of sleep apnea and other conditions causing breathing issues throughout the night.
- What Is Sleep Apnea?
- Recalled Philips CPAP Machines
- What is Polyester-Based Polyurethane (PE-PUR) Sound Abatement Foam?
- What is the off-gassing of diethylene glycol (DEG)?
- Side Effects of Exposure to PE-PUR Sound Abatement Foam
- Dangers of Silicone-Based Replacement for the PE-PUR Foam
- What to Do if You Have Been Using the Recalled CPAP Machines
- What if My Doctor Deems It Necessary to Continue Using a CPAP Machine?
- What Do I Do if My CPAP Machine Is Part of the Recall?
- How Does “Repair and Replace” Work?
- Report Your Side Effects Using an FDA Medwatch Voluntary Report Form
- Alternatives to Sleep Apnea CPAP Treatment
- Recent Update to the Philips Recall
- Process to File a Claim for Patients Affected by Philips’ Recalled CPAP Devices
- How Do I Prove My Cancer or Illness Resulted From Using a Philips CPAP Machine?
- Can Levin & Perconti Help Me Obtain Compensation for My Injuries Resulting From a Defective Philips CPAP Machine?
- Testimonials
The continuous positive airway pressure (CPAP) machines create a channel of forced air to keep airways open when sleeping. Most CPAP users have obstructive sleep apnea. However, researchers found that a sound-deadening foam in the machines used to silence its loud vibrations would break down, releasing harmful chemicals into users’ airways. Prolonged exposure to these chemicals can cause lung cancer and other health conditions.
What Is Sleep Apnea?
Sleep apnea is a condition that interferes with nighttime breathing. It can be either obstructive sleep apnea (the most common type), occurring when the throat muscles relax, narrowing or closing off your airway, or central sleep apnea, resulting from improper or a lack of signals from the brain to the muscles that control breathing. In some instances, patients can suffer from both types.
Patients afflicted by this sleep disorder stop breathing several times each night. You might wake up suddenly several times during the night and repeatedly gasp for air, or you might simply have trouble staying asleep. Morning headaches and dry mouth are also sometimes indicative of improper breathing during sleep. If you snore loudly or regularly feel tired after a full night’s sleep, you might have sleep apnea.
It’s important to speak with your doctor about possible sleep disorders if you suffer from any signs or symptoms of disrupted or inadequate sleep. Sleep apnea can be a potentially serious and dangerous condition. Complications of untreated sleep apnea include:
- High blood pressure
- Glaucoma
- Heart and cardiovascular problems, including heart attack
- Cancer
- Type 2 diabetes
- Metabolic disorders affecting weight, cholesterol, blood pressure, and blood sugar levels
- Cognitive impairment, affecting your ability to drive (e.g., drowsy driving) or perform other routine tasks
- Behavioral disorders
- Medication and general anesthesia complications (your health care providers may need to take extra precautions during surgery)
- Liver problems
- Sleep deprivation for you and your family
CPAP machines are a popular treatment for sleep apnea. These medical devices release air pressure into the sleeper’s nose, gently forcing their passages open to keep oxygen moving through the airways and into the lungs to provide adequate rest.
Common side effects of CPAP machine usage include dry mouth and eyes, congestion or runny nose, and nosebleeds. If you experience more severe side effects, such as stomach discomfort, bloating, or anything else unusual, you should discontinue use and contact your doctor immediately. Never alter your treatment regimen without consulting with your doctor.
Recalled Philips CPAP Machines
Philips issued a voluntary recall affecting several of its CPAPs, BiPAPs, and ventilators. If your device is on this list, keep reading for directions on how to get your machine repaired or replaced, or contact your doctor for more information about your continued treatment plan.
Philips Respironics recalled devices include:
- A-Series BiPAP A30
- A-Series BiPAP A40 (ventilator)
- A-Series BiPAP Hybrid A30
- A-Series BiPAP V30 Auto (ventilator)
- C-Series ASV (ventilator)
- C-Series S/T and AVAPS
- DreamStation
- DreamStation ASV
- DreamStation Go
- DreamStation ST, AVAPS
- Dorma 400
- Dorma 500
- E30
- Garbin Plus, Aeris, LifeVent (ventilator)
- OmniLab Advanced+
- REMstar SE Auto
- SystemOne ASV4
- SystemOne (Q-Series)
- Trilogy 100 (ventilator)
- Trilogy 200 (ventilator)
What is Polyester-Based Polyurethane (PE-PUR) Sound Abatement Foam?
The use of polyester-based polyurethane (PE-PUR) sound abatement foam in many home and industrial devices is common. It essentially acts as a replacement for rubber and helps to insulate or provide a “cushion” in many products. Likewise, Philips Respironics used this type of foam in its CPAP machines to reduce noise and vibrations made by the device.
However, recently, researchers found PE-PUR can cause health risks and lead to long-term health issues. The science behind the potential health risks to patients hinges on a design flaw that can lead to foam degradation, resulting in black particles entering the air tube, which users unknowingly inhale or ingest. Researchers have found these particles and other gasses released during the degradation of silicone, this particular foam to be potentially toxic. It may even possibly contribute to various cancers due to additives such as flame retardants.
Additionally, researchers have linked isocyanates used in PE-PUR manufacturing to severe respiratory problems, including shortness of breath, pneumonia, life-threatening asthma, pulmonary fibrosis, and bronchitis. The damage to the lungs can be permanent, with cancer and other health complications also apparent.
What is the off-gassing of diethylene glycol (DEG)?
As the foam in the recalled Philips CPAP devices ages, it releases Volatile Organic Compounds (VOCs). VOCs are chemicals released from many plastics and other human-made compounds. As chemists and doctors study these materials, they’ve found both short- and long-term adverse health effects associated with their use and human exposure.
Diethylene glycol (DEG) is one chemical off-gassing from the PE-PUR foam previously used in Philips Respironics CPAP machines, as it degrades. It’s a highly toxic chemical with exposure likely to cause many health problems in individuals, ranging from nausea and vomiting to coma and death.
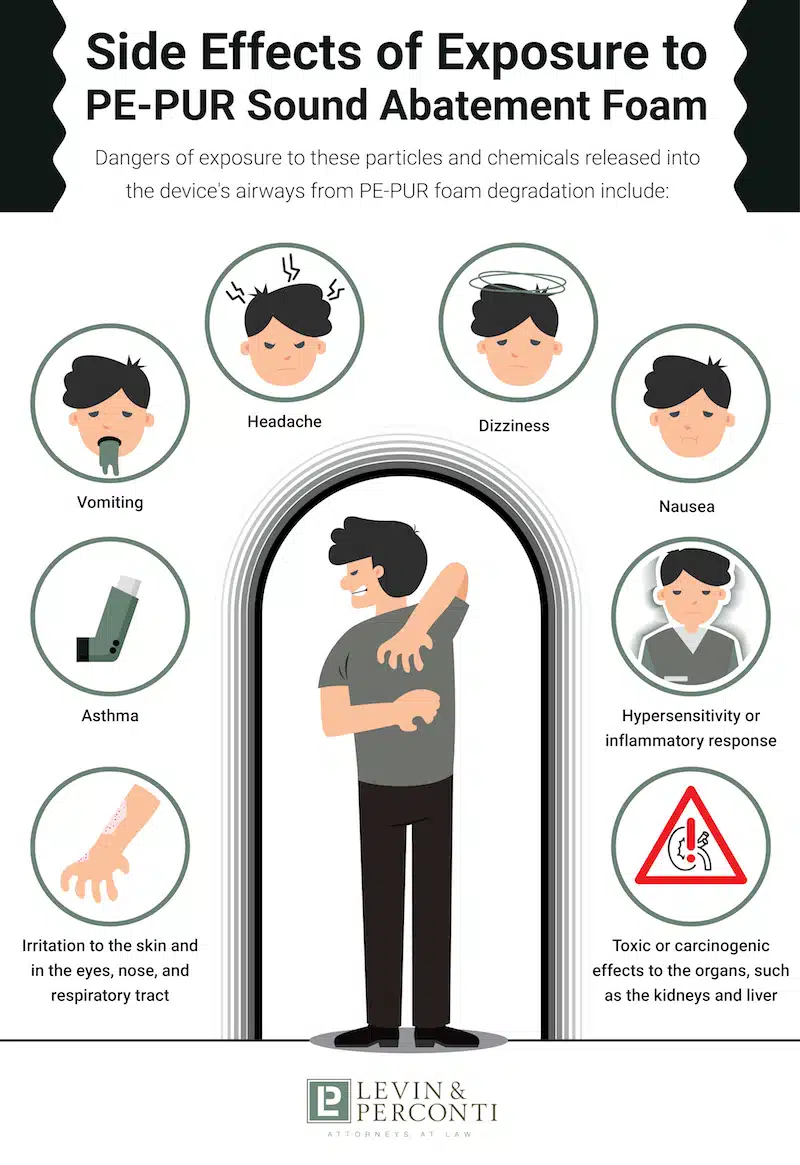
Side Effects of Exposure to PE-PUR Sound Abatement Foam
The consequences can be severe, permanent, or deadly when patients breathe in the chemicals or swallow the broken down particles from degraded polyester-based polyurethane (PE-PUR) sound abatement foam in Philips CPAP machines. The risk of foam degradation is higher when high heat and humidity is present, and when the device is cleaned improperly.
In its June 2021 recall notice, the FDA noted several complaints from users received by Philips Respironics, citing “reports of headache, upper airway irritation, cough, chest pressure, and sinus infection.” These symptoms began after patients became aware of black particles and debris in the devices’ air pathways.
Dangers of exposure to these particles and chemicals released into the device’s airways from PE-PUR foam degradation include:
- Headache
- Dizziness
- Irritation to the skin and in the eyes, nose, and respiratory tract
- Hypersensitivity or inflammatory response
- Nausea
- Vomiting
- Asthma
- Toxic or carcinogenic effects to the organs, such as the kidneys and liver
Dangers of Silicone-Based Replacement for the PE-PUR Foam
Philips is replacing the PE-PUR foam used in its CPAP devices with silicone-based foam. The silicone product also off-gasses volatile organic compounds (VOCs). These gas emissions can cause short and long-term health complications that mirror those seen with particulate and chemical exposure from PE-PUR foam degradation.
After conducting its own testing, Philips said the product was safe, thereby gaining FDA approval for the replacement foam. However, during the manufacturing facility inspection, FDA officials became aware of information not previously made available about possible safety issues with the new sound-abatement material. A singular, similar device manufactured outside the United States, containing the same silicone-based product, failed a safety test for VOCs, exposing a potential weakness in the replacement CPAP machines that could cause similar problems for patients injured by the PE-PUR foam.
The FDA is aware that many patients are already using the replacement devices containing the silicone-based foam and doesn’t have enough information at this time to conclude the risks associated with the same. The federal agency is requiring Philips Respironics to conduct further testing at an independent laboratory to confirm the replacement foam’s safety.
Until such a time as the FDA releases data regarding the additional evaluation of the product, there’s no recommendation to discontinue the use of the replacement CPAP or other ventilator machines. Patients should follow the advice of their health care team about the best course of action that aligns with their individual treatment plans.
What to Do if You Have Been Using the Recalled CPAP Machines
The FDA recommends you stop using the affected machines immediately. Visit your doctor and find a different CPAP or ventilator device to use or discuss alternative treatment options if relevant to your medical care plan.
Philips has announced that it will take back all the affected devices to repair or replace them. Talk to your doctor about the best course of action for you.
What if My Doctor Deems It Necessary to Continue Using a CPAP Machine?
Continue using your CPAP device until you receive a replacement or alternative treatment plan if your health care provider determines that the benefits outweigh the risks identified in the recall notification. Never stop or change your course of treatment without first speaking with your doctor. If you continue to use your machine, don’t use ozone cleaners and avoid storing it in high heat or humid environments. The FDA has issued a warning that these cleaners and environmental conditions can break down the foam at a faster rate.
You can get an inline bacterial filter to catch the small particles. This filter will not catch the outgassing chemicals and needs regular inspections. Talk to your doctor about other precautions.
What Do I Do if My CPAP Machine Is Part of the Recall?
Register your device with Philips Respironics by phone at 877-907-7508 or online. Visit your doctor if you’re experiencing any signs or symptoms of particulate or chemical exposure from PE-PUR sound-abatement foam degradation. You should also contact an attorney to discuss possible compensation for your injuries.
How Does “Repair and Replace” Work?
After you register your CPAP or another device on the Philips Respironics Device Recall Notification website, Philips will tell you how to send in your device for repair or replacement. For some customers, Philips will send a repair kit and a payment for the hourly wage of repairing it. Replacement and repair may take up to one year. Talk to your doctor about finding replacement respiratory care devices so that you can continue your recommended or required treatment.
Report Your Side Effects Using an FDA Medwatch Voluntary Report Form
If you experience particulate or chemical exposure symptoms or suffer other injuries after using a Philips Respironics CPAP device, use the FDA’s MedWatch Voluntary Reporting form to let the agency know what you’re experiencing. Voluntary reporting helps the FDA track unknown risks for approved health care devices and medications. Your doctor or other health care provider can also report problems with your device to the regulatory agency using a separate form.
Alternatives to Sleep Apnea CPAP Treatment
There are several alternatives to using a CPAP machine to treat sleep apnea. It’s important to seek medical advice from your doctor or other health care provider before changing your treatment.
Examples of sleep apnea treatment alternatives include:
- A device that warns when you move into a dangerous position that can impair your breathing
- Devices that hold the airways in your nose open to allow for adequate, continuous airflow
- An oral appliance that places your mouth and tongue better to prevent obstructions from relaxed throat muscles
- Nerve and tongue stimulation
$3 million
Settlement
against a car manufacturer that improperly designed the fuel delivery system of a vehicle, resulting in a fuel-fed fire after a head-on collision which killed the occupants of the vehicle.
$1.25 million
Settlement
for a 70-year-old patient who, as a result of treatment for prostate cancer, suffered radiation burns to the rectal wall due to improper placement of radioactive seeds (brachytherapy) in the prostate requiring numerous surgeries and loss of all bladder and bowel function.
$650,000
Settlement
against Rush Presbyterian and a doctor who administered a 17-year-old plaintiff brachytherapy for a cancerous tumor on the back of her leg and during the brachytherapy caused serious burns which took two years to heal.
Recent Update to the Philips Recall
As of April 2025, there are 826 ongoing cases in multidistrict litigation over injury claims related to the CPAP machines.
On April 29, 2024, Philips Respironics reached a $1.1 billion settlement regarding lawsuits tied to its CPAP devices. The settlement allocates $1.075 billion to personal injury claims from individuals who suffered serious health issues or death due to the recalled machines. An additional $25 million will fund medical monitoring for users who may face future complications.
The settlement is still awaiting approval from a federal court. If the settlement is not approved, bellwether trials for lawsuits alleging cancer and other health problems from the devices could begin in 2025. These federal cases will be overseen by Senior U.S. District Judge Joy Flowers Conti.
The FDA released the following statement in April 2024:
On April 9, 2024, a federal district court entered a consent decree against Philips Respironics (Philips). The consent decree includes key provisions aimed to prioritize patient relief and ensure the company’s regulatory compliance. The consent decree comes after Philips recalled certain ventilators, continuous positive airway pressure (CPAP) machines, and bi-level positive airway pressure (BiPAP) machines in June 2021 because of potential health risks—impacting 15 million devices worldwide. Patients impacted by the Philips recall remain a top priority for the agency as the FDA continues to take steps to protect the health and safety of individuals using these devices. – FDA.gov
In October 2021, a person suing Philips for the harms caused by its CPAP and other mechanical ventilator devices registered a class-action product liability lawsuit in Pennsylvania. Also that month, SoClean, Inc. filed a lawsuit against Philips Respironics for false and misleading statements. In November 2021, “following further assessment of the issue, and discussions with regulatory agencies,” Philips updated its recall notice.
Philips knew about the problem with the PE-PUR foam since 2016. The company failed to warn consumers and health care providers and chose to do nothing to correct the problem until FDA inspectors became aware of it. Philips received over 222,000 complaints that included the keywords “contaminants, particles, foam, debris, airway, particulate, airpath, and black.”
Process to File a Claim for Patients Affected by Philips' Recalled CPAP Devices
Contact an attorney and seek legal advice if you’ve suffered harm after using a Philips CPAP machine. An attorney can explain your rights, determine if you have a valid case, and help you decide what your next steps should be. The PE-PUR used in Philips CPAP and other mechanical ventilator devices can cause serious injuries, and it’s important that the responsible parties are held accountable and that you get the compensation you need to aid in your recovery.
CPAP lawsuits are still in the early phases. Several individuals have filed lawsuits in federal courts, and many are joining together in multidistrict litigation. Nearly 140 injured patients are seeking financial compensation for harms caused by Philips sleep apnea devices and mechanical ventilators in a Pennsylvania court (MDL-3014).
How Do I Prove My Cancer or Illness Resulted From Using a Philips CPAP Machine?
Your legal team can guide you in gathering evidence, including medical records and doctor’s notes, to prove your case. Save all your relevant health care information, medical bills, and receipts. Additionally, a lawyer can hire expert witnesses to establish causation.
Can Levin & Perconti Help Me Obtain Compensation for My Injuries Resulting From a Defective Philips CPAP Machine?
If the early cases are successful, others might be eligible to receive compensation for their damages. These damages may include expenses for medical bills, replacement machines, lost wages, or non-monetary losses, such as loss of enjoyment of life, pain and suffering, and possibly punitive damages if the court finds that Philips acted intentionally or recklessly without regard for others’ safety and well-being. Additionally, early verdicts may result in later cases settling out of court.
Settlements are more likely than verdicts in cases like this, but the litigation process can still be lengthy. It’s important to contact an attorney as soon as possible if you own one of the affected Philips machines and have suffered injuries. The attorneys at Levin & Perconti can give you a free case review to see if you’re a qualified candidate for a lawsuit.
Levin and Perconti have experience representing plaintiffs in personal injury and wrongful death cases. They’ve also represented injured people in product liability cases similar to Philips CPAP litigation. With something as important as your health or life, or the life of those you love, you need to know it’s in good hands. Levin & Perconti has an extensive history of delivering satisfactory results. Many of their past clients can attest to their level of compassion, dedication, and hard work. Contact Levin & Perconti today to see how they can best help you.
Testimonials
Not only were they so professional but also so caring and thoughtful. It was very difficult going over the facts in our mother's case but they were so compassionate and understanding and allowed us to be with them every step of the way. We were able to sit in on the depositions and we were really able to see how hard they worked on our behalf.
During that time, my family and I suffered the loss of my mother. Mr. Levin and Mr. Perconti were more than just our lawyers. Words can’t really express our gratitude.
They were most patient and responded timely to many questions throughout the entire process. All demonstrated professionalism and extensive knowledge of case and state laws. Their in-depth investigative work uncovered details which were unknown to us and yet sadly confirmed our suspicions. We would recommend the Levin and Perconti Law firm and especially this team.
During that time, my family and I suffered the loss of my mother. Mr. Levin and Mr. Peconti were more than just our lawyers. Words can’t really express our gratitude. My family and I will always be grateful for the compassion that they showed us. I want to thank you for what you did for my family, and for always being there for us.
Margaret’s representation, guidance, and professionalism gave us the sense that we could put our trust in Levin & Perconti and the final outcome proved that to be true.
Contact Us
- Free Consultation
- (312) 332-2872
"*" indicates required fields
Related Pages
- Product Liability Lawyer
- CPAP Machine Recall
- CPAP Machine Side Effects
- CPAP Machine Cancer Risk
- CPAP Machine Lawsuits
- Darvon and Darvocet Recall
- Spinal Steroid Injection Recall
- Testosterone Therapy Lawsuit
- Stryker Hip Recall Lawsuit
- DePuy ASR Hip Implants Recall
- Exactech Knee & Ankle Recall
- Defective Medical Devices
- L’Oreal Hair Straightening Uterine Cancer Lawsuits
Notable Results
SETTLEMENT
against a car manufacturer that improperly designed the fuel delivery system of a vehicle, resulting in a fuel-fed fire after a head-on collision which killed the occupants of the vehicle.
SETTLEMENT
for a 70-year-old patient who, as a result of treatment for prostate cancer, suffered radiation burns to the rectal wall due to improper placement of radioactive seeds (brachytherapy) in the prostate requiring numerous surgeries and loss of all bladder and bowel function.
VERDICT
against Rush Presbyterian and a doctor who administered a 17-year-old plaintiff brachytherapy for a cancerous tumor on the back of her leg and during the brachytherapy caused serious burns which took two years to heal.
Our Office
325 N LaSalle Dr Suite 300
Chicago, IL 60654
312-332-2872